Table of Contents
The problem that nobody tells at the beginning
Imagine this: you’ve been working on implementing your quality management system for six months. You have a consultant, you have the procedures written, you have the quality policy approved by management. But when you search for the updated version of the purchasing procedure, there are three different files on Google Drive with almost identical names. The internal auditor asks for last quarter’s training records and nobody knows exactly where they are. And the external audit is in six weeks.
This situation is not an extreme case. It is the reality of most SMEs when they try to implement ISO 9001 without a clear structure for managing system information.
The standard itself is not the obstacle. The problem is that no one teaches you how to manage the system as you build it.
This guide is written for exactly that moment: when you’ve decided you want certification (or just a functional QMS), and you need to understand what steps to take, in what order, and what pitfalls to avoid.
What is really ISO 9001 and what is not?
ISO 9001 is the international standard that establishes the requirements for a quality management system. It is published by the International Organization for Standardization (ISO) and its current version is ISO 9001 for SMEs:2015.
What the standard asks of you, in concrete terms, is that:
- Understand the context of your organization: who are your customers, what external and internal factors affect your business, who are your stakeholders.
- Define processes: how you do what you do, with clear responsibilities and measurable criteria.
- Control your documents and records: that the information is updated, available and traceable.
- Measure and improve: indicators, internal audits, management review, corrective actions.
What ISO 9001 is not:
- It’s not a guarantee that your products will be perfect.
- It is not an operations manual that dictates how to work in detail.
- It is not exclusive to large companies or specific industries.
- It does not require you to have a quality department with ten people.
This is important to understand from the beginning: the standard gives you a framework, not a recipe. You decide how to apply it according to the size and reality of your company.
Is it worth it for an SME? The honest question
Let’s be blunt: implementing ISO 9001 for SMEs has a real cost – in time, money and organizational effort. Before you commit, it pays to do the math with your eyes open.
When does it make sense
- Your company has customers or tenders that require certification as an entry requirement.
- You are growing and informal processes no longer scale: errors are repeated, coordination fails, training is inconsistent.
- You want to expand into international markets where ISO 9001 is a sign of credibility.
- You already have some order in your processes and want to formalize them in a structured way.
When can you expect
- Your company has less than 10 employees and the processes are completely handcrafted – implementing a formal QMS at that time may generate more bureaucracy than real benefit.
- You don’t have the minimum availability of time in the management team. Without management commitment, a QMS does not work.
- You seek certification just for marketing, with no intention of keeping the system alive.
If you’re not sure where you stand, the free ISO 9001 Feasibility Test for SMEs takes less than five minutes and gives you an objective reading of whether your company is ready to get started.
How to implement ISO 9001 for SMEs: the actual stages
There is no single universal path, but most successful SME implementations follow a logical sequence similar to this one. Times are estimates and vary greatly depending on the size of the company, the availability of the team and whether you work with an external consultant or not.
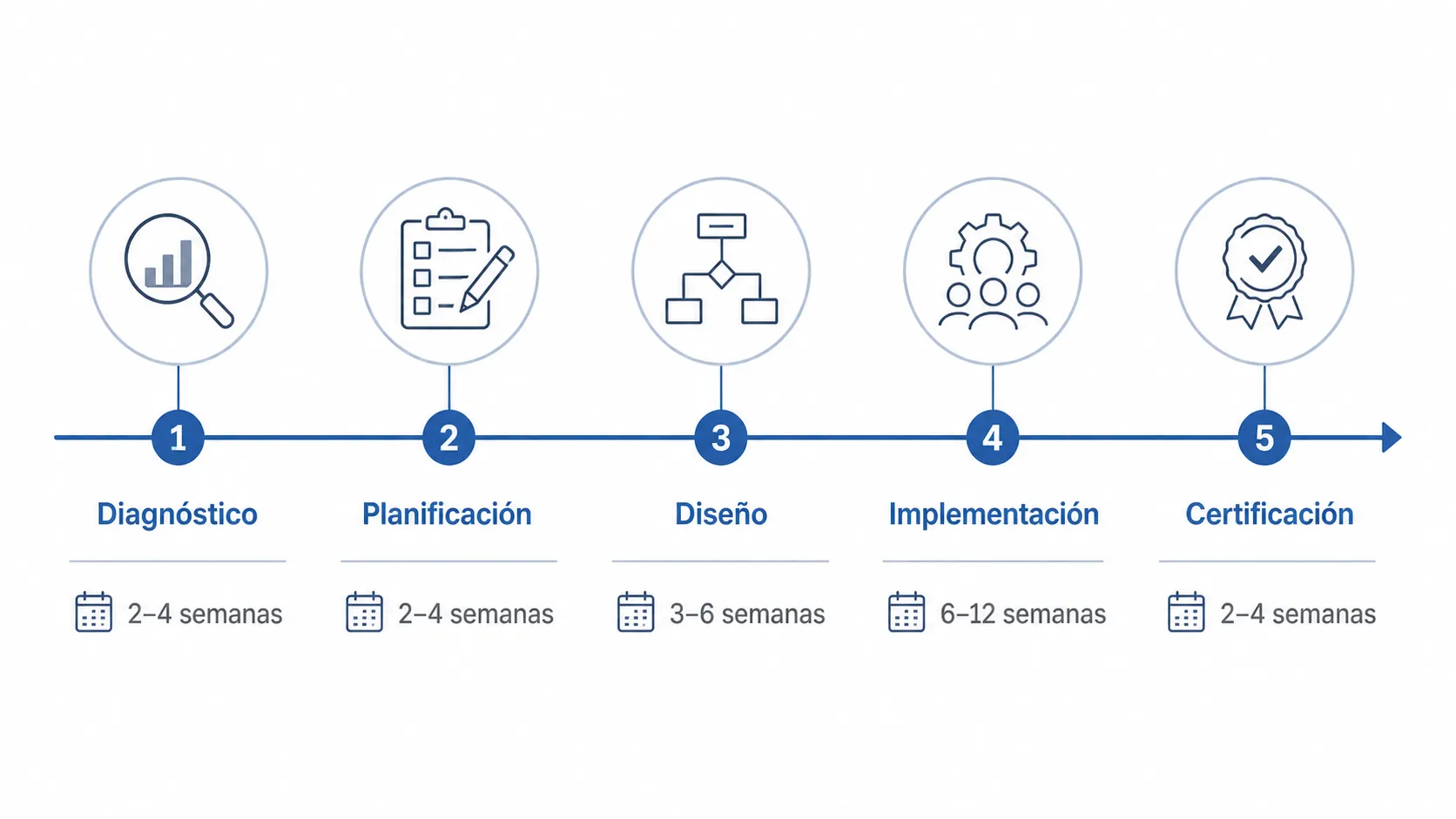
Stage 1 – Initial diagnosis (2-4 weeks)
Before designing anything, you need to know where you are. This involves:
- Mapping the company’s current processes (the real ones, not the ones you think exist).
- Identify what documentation you already have and what state it is in.
- Assess the gap between your current situation and the requirements of the standard.
The result of this stage is a gap analysis that tells you, in concrete terms, how much work you have ahead of you.
Useful resource: the QMS Maturity Diagnosis helps you to do this analysis in a structured way.
Stage 2 – Project planning (1-2 weeks)
With the diagnosis in hand, you define:
- QMS scope: which processes and areas the system includes (and which it does not).
- Realistic schedule with milestones and responsible parties.
- Resources needed: internal team time, external consultant if applicable, management tools.
This stage is brief but critical. Many implementations fail because they were launched without a clear plan.
Stage 3 – System Design (2-4 months)
The longest stage. Here you develop the content of the QMS:
- Organizational context: analysis of the environment, stakeholders, formal scope of the system.
- Quality policy and objectives: what you are committed to achieve and how you will measure it.
- Map of processes and procedures: how each process works, who is responsible, what records it generates.
- Supporting documentation: formats, instructions, acceptance criteria.
A common mistake at this stage: documenting how everything should work, rather than how it actually works. Auditors are quick to detect when documentation does not reflect actual practice.
Stage 4 – Implementation and operation (2-3 months)
Documentation alone is worthless if it is not used. In this stage:
- You train staff on the new procedures.
- You start generating the records that the system requires.
- You conduct the first internal audit: a systematic review that the system is working as planned.
- You conduct the first management review: top management evaluates the system’s performance and makes decisions.
Stage 5 – Certification audit (variable)
If you seek formal certification, you hire an accredited certifying body. The typical process has two phases:
- Stage 1 (documentary)audit: the auditor reviews that your documentation meets the requirements of the standard.
- Stage 2 (on-site)audit: verification that the system is implemented and working in practice.
If there are non-conformities, you have a deadline to resolve them before the certificate is issued.
| Stage | Estimated duration | Main Responsible |
|---|---|---|
| Initial diagnosis | 2-4 weeks | Quality manager |
| Planning | 1-2 weeks | Management + quality manager |
| System design | 2-4 months | Quality Manager (with or without consultant) |
| Implementation and operation | 2-3 months | The whole team |
| Certification audit | 1-4 weeks | External certifying body |
Total estimated time for an SME: 6-12 months from zero to certification. Companies with documented processes can do it in less time.
The most common mistakes (and how not to fall into them)
After seeing many implementation processes for SMEs, there are patterns that are almost always repeated when something goes wrong.
1. Document for compliance, not for use.
The most common mistake. Extensive and detailed procedures are created that no one actually reads. The result: a huge gap between what the system says and what the team does. Auditors spot it immediately.
How to avoid it: Document just enough. Prioritize clarity and practicality over length and formality.
2. Underestimate the real time required by the QMS.
Implementing ISO 9001 for SMEs is not a weekend project. In an SME of 50 people, the quality manager may spend between 30% and 50% of his time on the project during the first few months.
How to avoid it: Be realistic with management about the effort involved. If the quality manager has other operational responsibilities, the project will take longer.
3. Do not involve management from the beginning.
ISO 9001:2015 explicitly emphasizes leadership. A QMS that management considers a “quality area thing” and not a business management system is unlikely to survive the first follow-up audit.
How to avoid it: Quality policy, objectives and management review are not formalities. They are the mechanism by which top management directs the system.
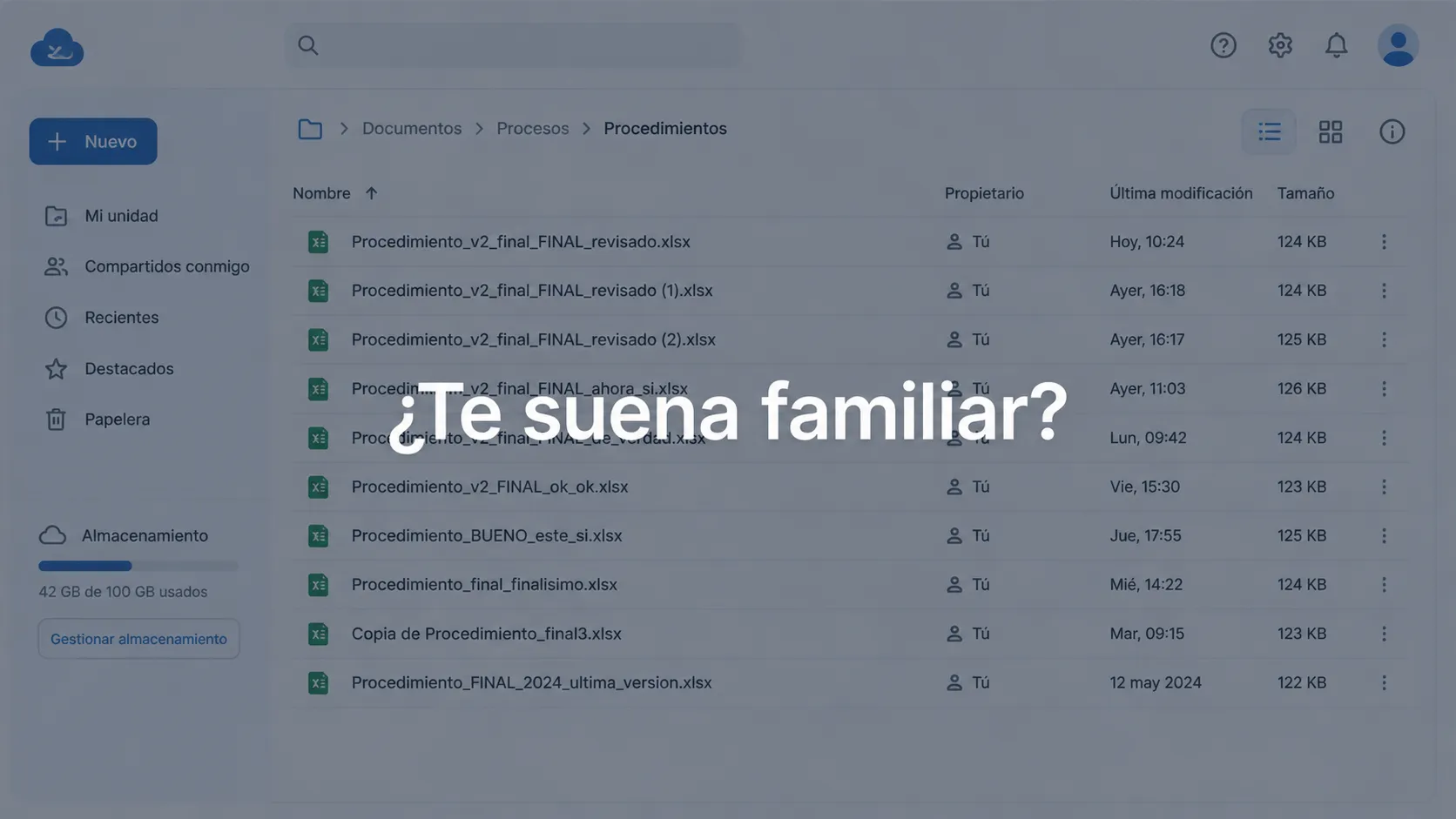
4. Manage the QMS in Excel and Google Drive without structure.
This deserves its own point because it is extremely frequent: the system is well designed, but it is managed in a shared folder that no one controls. Documents without version, records that get lost, formats that everyone modifies on their own.
How to avoid it: Define from the beginning how you are going to manage the system’s information – where documents live, who can modify them, how versions are managed. Tools like QualityWeb 360 are designed specifically for this.
5. Get certified and leave the system
Certification is not the end of the project. It is the beginning of maintenance. Many companies obtain certification and drastically reduce the effort on the QMS, with the result that the next follow-up audit (usually annual) finds the system deteriorated.
How to avoid this: Design the QMS to be sustainable with the actual level of effort your team can maintain. A simple system that works is always better than an elaborate one that no one sustains.
Watt you need to manage your QMS without going crazy
Here’s something that implementation books often don’t make clear: designing the system and managing it are two different activities.
You can do a great job designing your processes, documenting your procedures and defining your metrics. But if you don’t have an orderly place to live that system day to day, chaos returns quickly.
The minimum you need to manage a functional QMS:
Document control with traceability Know what version of each document is current, who approved it and when. Without this, any audit becomes a treasure hunt.
Every activity that the standard requires to be recorded – training, calibrations, audits, reviews – needsa place where the evidence is available and retrievable.
Tracking nonconformances and corrective actions The heart of continuous improvement. If you don’t systematically track what went wrong and what you did about it, the system doesn’t improve.
Visible indicators Quality objectives need to be measured. If KPIs live in a spreadsheet that no one updates, they are no longer useful.
Many SMEs start with Excel and Google Drive, and it makes sense: they are readily available and familiar tools. The problem comes when the system grows – more documents, more records, more people involved – and those tools are not designed to reliably manage versions, permissions and traceability.
If you want an objective reading of where your QMS stands today, the QMS Maturity Diagnosis is a good place to start.
Frequently Asked Questions
How much does it cost to implement ISO 9001 in an SME?
The cost varies significantly depending on whether you hire an external consultant or do it with internal resources, the size of your company and the certifying body you choose. Generally speaking, an implementation with a consultant for an SME of 20-50 people can be between USD 5,000 and USD 20,000, including the certification audit. Without a consultant, the direct cost drops considerably, but the internal team time is the real cost. Accredited bodies such as ICONTEC, Bureau Veritas or SGS publish their certification fees for different company sizes.
Can I implement ISO 9001 without hiring a consultant?
Yes, it is possible. The standard does not require the hiring of a consultant. Companies with a quality manager with experience and available time can carry out the implementation internally. The key is to have a good understanding of the requirements of the standard, access to appropriate training and tools to facilitate the administration of the system. That said, a consultant with experience in your industry can significantly shorten implementation time and help you avoid frequent mistakes.
How long does certification take once implementation is complete?
Once the system is implemented and operating (with at least one full cycle of internal audit and management review), the actual certification audit can take anywhere from one week to one month, depending on the size of the company and the availability of the certifying body. Typically, the stage 1 (documentary) and stage 2 (on-site) audits are separated by two to four weeks.
Does ISO 9001 apply only to manufacturing?
No. ISO 9001 is a generic standard that applies to any type of organization, regardless of its industry or size. It is widely used in services, logistics, healthcare, technology, food, construction and many other sectors. The standard does not prescribe how you should do things – you adapt the requirements to the reality of your business.
What happens if my company does not pass the certification audit?
If the auditor finds non-conformities, it does not automatically mean you lose certification – it depends on the severity. Minor non-conformities generally have a timeframe (60 to 90 days) to resolve with documented evidence. Major nonconformities may require an additional audit. The important point is that the audit process is a review, not an examination without the possibility of correction. Most companies that come to the audit having seriously implemented the system pass the audit, although almost always with observations for improvement.
Is your company ready to take the plunge?
Before hiring a consultant, buying software or booking a date with the certifier, it’s worth taking five minutes to do an honest reading of your current situation.
The ISO 9001 Feasibility Test is free, takes less than five minutes and gives you an objective assessment of whether your company has the conditions to begin a successful implementation – or what you need to resolve first.
No sales, no mandatory registration. Just one useful answer.